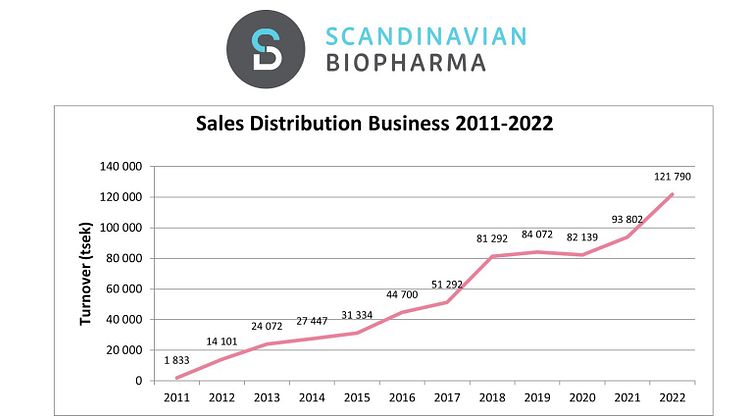
Scandinavian Biopharma presents the year-end report for 2022 with a 30% growth, record sales and the ETEC vaccine candidate about to enter phase 3
It is with great pleasure that we present another really good year of growth. The figures for the full year 2022 add up to an increase in sales equivalent to 30% in organic growth. In total, sales amount to approximately SEK 122 million. Our ETEC vaccine candidate, ETVAX, is about to enter phase 3.